Blood Products Regulations In India Presentation
| Introduction to Blood Products Regulations in India | ||
|---|---|---|
| Blood products regulations in India are governed by the Drugs and Cosmetics Act of 1940. The Central Drugs Standard Control Organization (CDSCO) is responsible for regulating and overseeing the manufacturing, import, and distribution of blood products. The objective of these regulations is to ensure the quality, safety, and efficacy of blood products in India. | ||
| 1 | ||
| Classification of Blood Products | ||
|---|---|---|
| Blood products in India are categorized into three main groups: whole blood, components, and derivatives. Whole blood refers to blood collected from a single donor, containing all its components. Components include packed red blood cells (PRBCs), platelet concentrates, fresh frozen plasma (FFP), and cryoprecipitate. | ||
| 2 | ||
| Licensing and Approval Process | ||
|---|---|---|
| Manufacturers of blood products in India must obtain a license from the CDSCO to operate. The licensing process involves submitting an application, providing manufacturing and quality control details, and undergoing inspections. Once approved, manufacturers must adhere to Good Manufacturing Practices (GMP) guidelines and obtain periodic renewals. | ||
| 3 | ||
| Quality Control and Testing | ||
|---|---|---|
| Blood products in India undergo rigorous quality control and testing procedures. The CDSCO mandates testing for infectious diseases, including HIV, hepatitis B and C, syphilis, and malaria. Quality control also includes ensuring proper storage conditions, labeling, and traceability of blood products. | ||
| 4 | ||
| Donor Screening and Blood Collection | ||
|---|---|---|
| Donor screening is a critical aspect of blood products regulations in India. Donors must undergo a thorough medical history and physical examination to determine their eligibility. Blood collection centers must follow strict protocols for aseptic techniques, equipment sterilization, and donor confidentiality. | ||
| 5 | ||
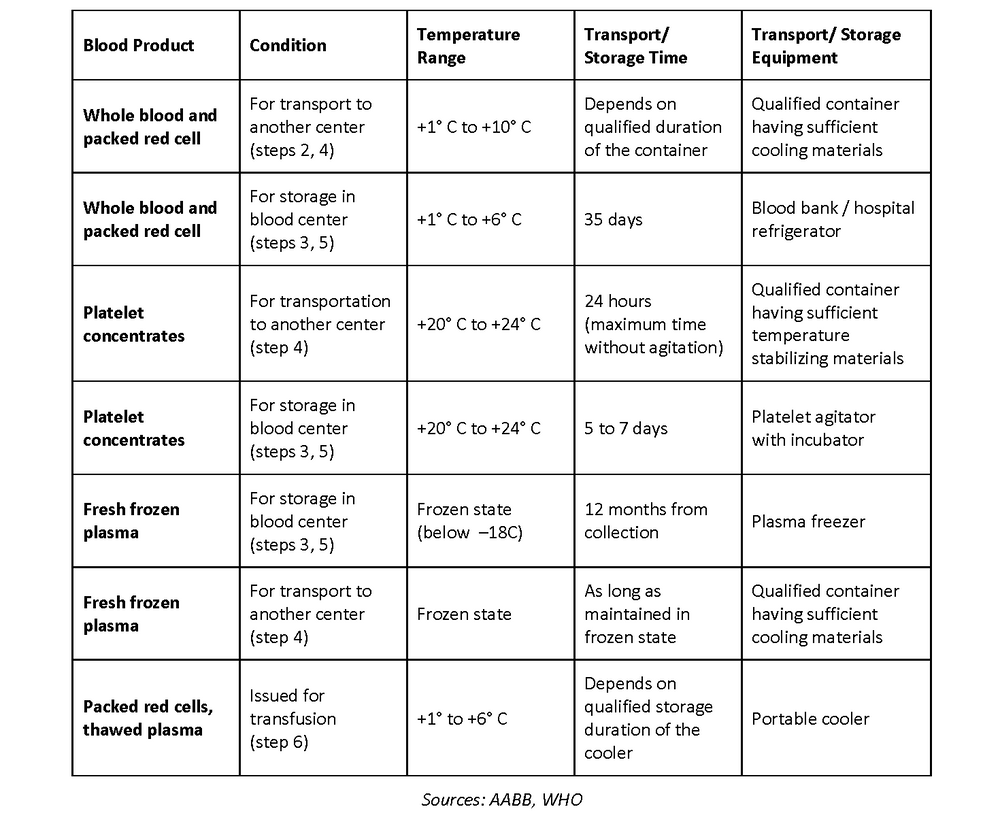
| Transportation and Storage Guidelines | ||
|---|---|---|
| Blood products must be transported and stored under controlled temperature and conditions. The CDSCO provides guidelines for temperature monitoring, proper packaging, and transportation vehicles. Storage facilities must have appropriate refrigeration systems, backup power supply, and emergency procedures in place. | ||
| 6 | ||
| Adverse Event Reporting | ||
|---|---|---|
| Adverse events related to blood products must be reported to the CDSCO within a specified timeframe. This includes any incidents of transfusion reactions, infections, or other serious adverse events. Reporting adverse events helps to identify potential risks and improve the safety of blood products. | ||
| 7 | ||
| Regulatory Inspections and Audits | ||
|---|---|---|
| The CDSCO conducts routine inspections and audits of blood product manufacturing facilities. Inspections ensure compliance with regulatory requirements, including GMP guidelines. Non-compliance can result in penalties, suspension of licenses, or even legal action. | ||
| 8 | ||
| International Collaborations | ||
|---|---|---|
| India collaborates with international organizations like the World Health Organization (WHO) and International Society of Blood Transfusion (ISBT) for blood product regulations. These collaborations help in sharing best practices, technical expertise, and harmonization of standards. India actively participates in global initiatives to improve blood safety and access. | ||
| 9 | ||
| Conclusion | ||
|---|---|---|
| Blood products regulations in India play a crucial role in ensuring the safety and availability of blood and its components. The CDSCO's oversight and enforcement of these regulations are essential for maintaining the quality of blood products. Continued efforts to strengthen regulations, improve donor screening, and enhance quality control will further enhance blood safety in India. | ||
| 10 | ||